VIVAN Life Sciences Offers wide range of Pharmaceutical Reference Standards & Pharmaceutical Characterized Standards. All the standards are compounds or chemical material of established quality and purity as shown by CoA, HPLC, MASS, and NMR quality analysis Reports, which conforms to Global quality standards.

VLST-02005
56392-14-4
C₁₄H₂₁NO₄
267.32

VLST-02006
21535-47-7
C₁₈H₂₁ClN₂
300.83

VLST-02007
83701-22-8
C₉H₁₅N₅O₄S
289.31

VLST-02008
85650-52-8
C₁₇H₁₉N₃
265.35

VLST-02009
82586-52-5
C₂₇H₃₅ClN₂O₇
535.03
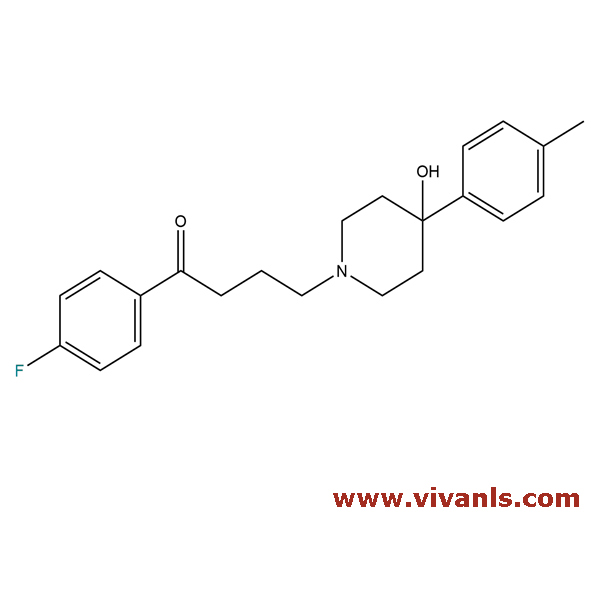
VLST-02010
1050-79-9
C₂₂H₂₆FNO₂
355.45
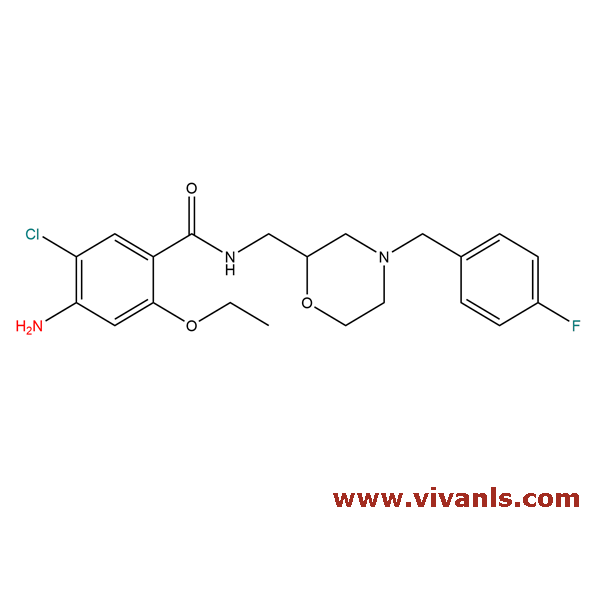
VLST-02011
112885-41-3
C₂₁H₂₅ClFN₃O₃
421.89

VLST-02012
192927-63-2
C₂₁H₂₇ClFN₃O₅
455.91

VLST-02015
389-08-2
C₁₂H₁₂N₂O₃
232.24

VLST-02016
235114-32-6
C₅₆H₇₁N₉O₂₃S
1270.27

VLST-02017
211914-51-1
C₂₅H₂₅N₇O₃
471.51

VLST-02018
75530-68-6
C₁₉H₁₉N₃O₆
385.37